Researchers Discover the Cell’s Secret Anti-Aging Mechanism
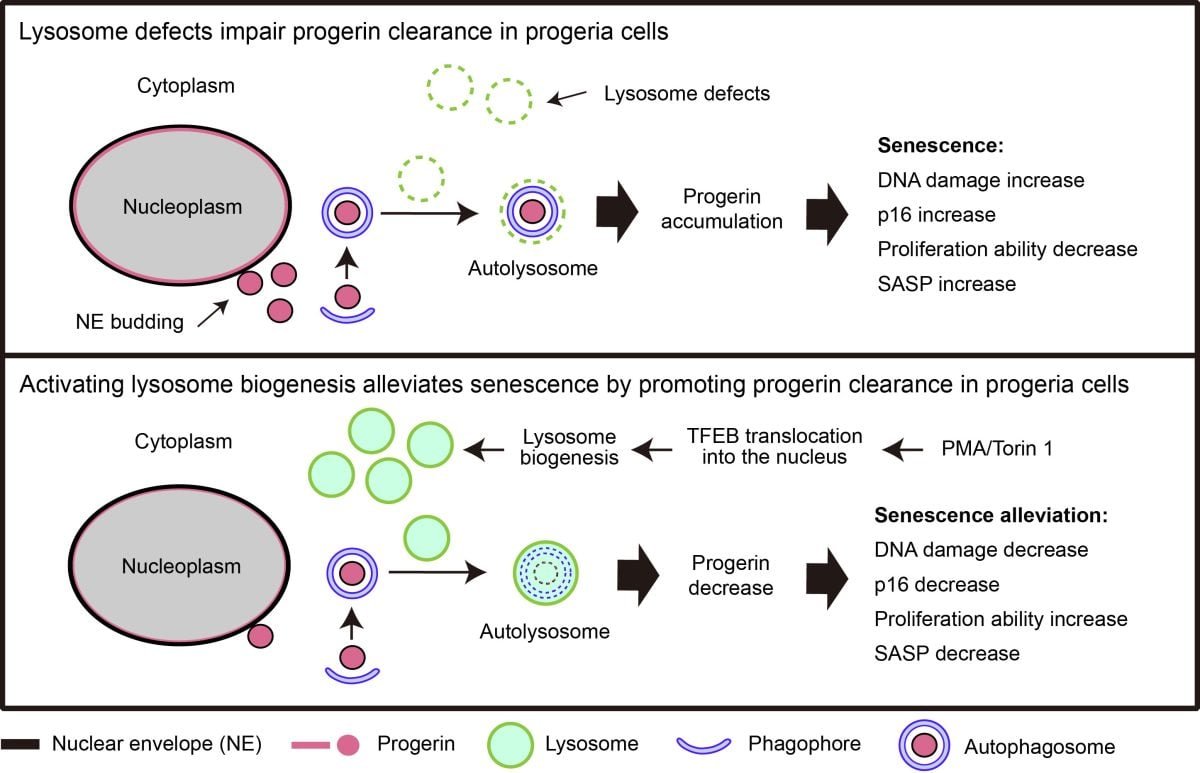
Activating lysosome biogenesis helps alleviate cellular senescence in progeria.
Hutchinson-Gilford progeria syndrome (HGPS) is a rare genetic condition that causes rapid aging and a range of visible and internal symptoms. Individuals with HGPS often experience early skin wrinkling, reduced skin elasticity, loss of body fat beneath the skin, hair loss, hardening of the arteries (atherosclerosis), and insulin resistance.
Research has shown that about 90% of HGPS cases are linked to a defective protein called progerin, which disrupts normal cell function. Progerin exerts a dominant-negative effect, leading to several cellular problems such as nuclear envelope (NE) deformation, increased DNA damage, telomere shortening, cell cycle arrest, and poor cell proliferation.
Recent studies have revealed that progerin is not limited to HGPS. It also appears in normal aging and chronic kidney disease (CKD), suggesting that it plays a broader role in age-related cellular decline. Because of this, improving the body’s ability to remove progerin has become an important focus for developing treatments targeting HGPS, CKD, and other diseases related to aging. A research team led by Professor Chuanmao Zhang from Peking University and Kunming University of Science and Technology has been studying the biological mechanisms behind both progeria and aging. In a new paper published in Science China Life Sciences, the group describes how progerin is cleared from cells through a lysosome-dependent pathway. Their work also identifies defects in lysosome function in HGPS and shows that enhancing lysosome biogenesis can repair these problems, promote progerin removal, and reduce cellular aging.
These discoveries highlight the crucial role of lysosomes in maintaining cellular health and eliminating harmful proteins. The study offers fresh perspectives on how stimulating lysosome activity could lead to new treatments for HGPS, CKD, and other age-related disorders. Tracking Progerin Through the Cell
Through immunofluorescence, live-cell imaging, and biochemical analyses, they discovered that the NE-localized progerin could be transported to the cytoplasm via NE budding and subsequently degraded through the autophagy-lysosome pathway. Further investigation revealed that progerin translocated to the cytoplasm was not efficiently cleared and accumulated in the cytoplasm.
To elucidate the underlying molecular mechanisms, RNA sequencing was performed on primary cells from two HGPS patients, which demonstrated significant downregulation of genes associated with lysosomal function in both patients. Lysosome defects in HGPS were further validated by reverse transcription quantitative PCR (RT-qPCR), immunofluorescence, and biochemical assays.
Subsequently, they explored whether counteracting lysosome defects could enhance progerin clearance and mitigate cellular senescence. Lysosome biogenesis was activated through two distinct approaches: stimulation of protein kinase C (PKC) or inhibition of mammalian target of rapamycin complex 1 (mTORC1). The results indicated that activation of lysosome biogenesis markedly counteracted lysosome defects, accelerated progerin clearance, and alleviated key senescence-associated phenotypes, including DNA damage, cell cycle arrest, and diminished proliferative capacity.
In conclusion, this study not only delineates the critical role of lysosomes in progerin clearance but also highlights the anti-aging potential of targeting lysosome biogenesis, offering innovative therapeutic avenues for HGPS and other age-related disorders.
